There is a lot of misinformation in the landscape architecture community about whether or not trees roots will grow in gravels, structural stone soils, and clear stone. Our experience indicates that roots will adventitiously grow in any medium given the right set of circumstances. Quantifying and predicting what set of circumstances produce that growth response is a whole different issue – the relative level of complexity in modeling this situation and overall difficulty in isolating variables make it difficult to predict accurately. We do know with relative certainty the general conditions and relative levels of functional needs for tree rooting in soils, but each tree species and individual is different in their response and timing.
When considering the functional needs of the tree in urban environments, this is the hierarchy of soils that trees will preferentially grow in, from best to worst:
1) Uncompacted native soil, by species, in suspended pavement system (such as Silva Cells)
2) Structural soils
3) Compacted base – clear stone (no fines)
4) Compacted base – aggregates with fines
Dr. Ed Gilman, a renowned professor of urban trees and landscape plants at the University of Florida, has done some excellent research about this issue. His 2006 study, “Deflecting roots near sidewalks,” is what I will be referring to primarily to examine this issue.
Summary of Gilman (2006) study and findings
Gilman’s (2006) study focused on examining the ways adventitious rooting in urban sidewalks could be averted using different materials. He varied his root barriers and soil types (well-drained and poorly-drained) while keeping other materials – sidewalk material type, depth, light conditions – consistent between plots, as well as having a control (no sidewalk plot or surface treatment) for comparison. Six types of root barrier treatments were used in the experiment:
#1 | Control plots – no sidewalk and no root barrier
#2 | Treatment : Sidewalk and no root barrier
#3 | Treatment : Biobarrier® 30cm (12”) D, installed vertically next to sidewalk, to 1” above final soil surface elev.
#4 | Treatment : DeepRoot® 30cm (12”) D, installed vertically next to sidewalk, to 1” above final soil surface elev.
#5 | Treatment : 6mm thick polyethylene, installed vertically next to sidewalk, to 1” above final soil surface elev.
#6 | Treatment : 15” depth – 1”ᴓ clear gravel spread over final surface with a front-end loader blade and then CIP concrete poured over surface of gravel, CIP not to penetrate gravel
Sycamore (Platanus occidentalis L.) trees were used in this study, and noted to be relatively fast growing trees that also produced a majority of root volume in the upper portion of the soil profile near to the surface. Similar stock (DBH and morphology was maintained between test subjects) was planted in each plot and then monitored for root growth throughout the 7-year term of study.
Gilman’s findings after the term of study were summarized as follows:
– Installation of a simple sidewalk section (#2, #3, #4, #5, #6) deflected roots to deeper soil layers
– The control plot (#1) yielded shallower average rooting depths than the sidewalk section with no root barrier (#2) in both soil types
– In well-drained soils:
- average root depth was significantly greater in sidewalks with a gravel sub-base (#6) than with other treatments (#2, #3, #4, and #5, not including the control (#1));
- average root depth was not variable among other root barrier treatments (#3, #4, #5); and,
- the average root depth was not significantly different for root barriers (#3, #4, #5) than for the sidewalk without root barrier (#2).
– In poorly-drained soils:
- Biobarrier (#3), gravel sub-base (#6), and the polyethylene (#5) performed equally well at directing roots away from the concrete
- the Biobarrier (#3) produced significantly deeper roots than other treatments, but the difference between these was indicated to be “statistically significant” but in all likelihood within the range of “real-world” variation between individuals and of negligible significance
– Root diameter was overall consistent/similar between all treatments and soil types.
The Gilman study also examined a horizontal application of gravel, with pavement above the gravel, so the trees had nothing to gain by growing through the gravel. Very little research has documented the effect of gravel as a vertical barrier between two rootable soil volumes.
The ability of tree roots to penetrate a vertical ballast layer will likely vary depending on the site. While ballast has been shown to reduce root growth in a horizontal application, tree biology suggests that if plentiful moisture, nutrients, and air are available on the other side of the gravel, tree roots may be able to penetrate the vertical ballast barrier. Soil suitable for tree growth will generally provide better resources for root growth than ballast, and we recommend using rootable tree soil whenever possible. But ballast may in some cases allow roots to bridge to nearby soil. In growing roots to access these nutrients, it would seem that the better the ability of soil nutrients to be provided during that growth response, the better the ability of the tree to benefit directly from and be sustained during the response. A native, uncompacted soil environment would provide the best environment for tree rooting and should be the primary option for best growth. When the soil is not a viable option in design or space is limited, we suggest working to minimize the bridging stone width between adjacent soil areas as much as possible.
Discussion of principles of tree root development in response to environmental stimuli
Although Gilman (2006) was a well-thought out and executed study, it is complicated to generalize and transfer the results to the all lateral tree root development in the rock-based soils. The use of a single chosen tree species (in Gilman’s study, Sycamore – Plantanus occidentalis) does not generate enough evidence to statistically-correlate the effects seen in the study with the larger interaction of all tree species. With regard to whether or not ballast (clear stone) will act as a barrier to lateral root movement, we offer the following information to guide the overall discussion.
Dr. Kim Coder (2007) summarizes tree root growth requirements in Table 1 below:
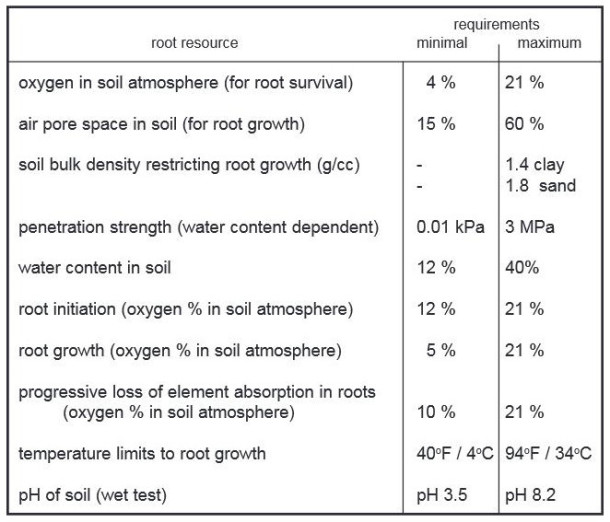
Table 1: Soil Based Root Growth Resource Requirements (Coder 2007)
According to Coder’s table, the minimum water content needed in soil for roots to grow is 12 percent. That being said, we have seen roots grow across gravel in order to reach resources beyond the gravel, and roots have even been seen to grow in bunkers (see Figure 1).

Figure 1: roots growing into a bunker (photo from City of Osnabruck).
People are sometimes passionate in their enmity toward tree roots that disturb paving or foundations, but trees are neither malicious in rooting nor cognizant of their impact on sidewalks (or the people who use them). A tree’s growth and development is entirely directed towards positive responses to environmental stimuli. If there is soil-available moisture, stomata open and photosynthesis initiates. If nutrients and moisture are available, roots grow to take advantage of those resources. If disease affects a portion of the tree, a compartmentalization response is generated, even to the point of abscission of the affected portion to retain overall viability of the individual. Satisfying these functional needs propels the growth and development of the tree via feedback loops with sensory information at various locations in the tree – roots, leaves, stems, etc. Depending on the information sent by each of these systems, a conditioned response is generated by the tree to maintain the status quo.
Looking at rooting, it is important to understand what constitutes a “positive” response by the tree, as seen by the growth of roots. Growth response triggers may be chemical, physical, or biological in nature. The ultimate goal of the rooting response and all production by the tree is based on a simple equation for expending the least amount of (internal) reserves/resources to capture the most (environmental) nutrient resources that will produce the most “success” for the individual. Among other abiotic and biotic factors, soil conditions play a large role in generating a positive outward rooting response. As their immediate soil environment is the only place that trees may capture the majority of their nutrients, it is critical that the strategic response by the tree is efficient and productive in yielding the desired results.
Lynch et al. (2012) indicate that the soil environment is extremely difficult to study due to complexity and non-uniformity at all scales and over time. Not only are abiotic factors such as nutrient availability and spatial locations of nutrient sinks not fixed, one must also consider the competition for these finite resources among all individuals – vegetative and animal. Additionally, the response of the individual tree may differ from the overall traits of the species due to environmental differences where the tree develops. For example, you may see the leaves of different individuals of the same species of tree be entirely different sizes, colors, or shapes in response to different soil conditions.
In the business world, you will sometimes hear the phrase “growth consumes capital.” The same principle can be applied to trees, where the risk and restructuring of vegetative rooting resources can produce great gains, but the “capital” that they consume in order to grow is stored sugars in the form of carbohydrates. The gamble of tree growth is similar to an economic model in that the growth response must be balanced with the potential for a positive result. A feedback system to either continue the response or alter it is based on maintaining the health of the tree.
So what does this all have to do with ballast and tree roots, anyhow? The answer lies in an equation that only the tree can answer in its development. In other words, in the thousands of years they have existed, trees have generated a set of typical responses to the environmental conditions with which they developed that is programmed in their DNA. These responses include everything from standard rooting depth to best capture nutrients, balancing growth responses with metabolic needs, calculated response to growth opportunities, the size of canopy to support photosynthetic processes, and so on. Whether ballast will act as a root barrier depends on the ability of the tree to react to the environmental stimuli of capturing nutrients in its environment inside of the barrier, balanced by the opportunity that the growth of the roots through the “barrier” to the adjacent area would offer for the resource capture and success of the tree.
Often, street trees are provided with such minimal soil volumes – far less than would be required to support it at maturity – that there will frequently be a growth response through the stone when the tree is large enough and there are excess reserves to support the effort. Lynch et al. (2012) indicate the basic positive response of root growth is complex and relatively unpredictable in timing, but can be attributed to the following factors in no particular order: soil chemical factors – P/N/Mg, pH, oxygen/aeration, organic material; soil organisims – pests and positive associations with bacteria; physical soil factors – mechanical impedance, water content, and temperature; and the shoot/root ratio, as an indicator of the general distance the tree needs to grow to capture nutrients (Lynch, Marschner and Rengel 2012; McMichael and Quisenberry 1993; Yau 2012).
Discussion of the role of different soils and structures in root development
Gilman et al. (1987) indicates rates and characteristics of root growth can be negatively affected or discouraged by soil-limited oxygen (chemical) and particle compaction (mechanical) factors. The factors are linked to the overall structure of the soil, specifically the soil macropores (the space created between individual soil particles). In relatively uncompacted soil, the particles are physically more separate from each other, as compared to a compacted soil of the same type. As soil particles become closer in proximity to neighboring particles, either via compaction or by migration of smaller soil particles such as silts or fines into larger pores, the oxygenated soil voids volume is reduced, thereby lowering the overall soil oxygen content available for root respiration and growth. Notably, the typical root appearance and character is affected by this limitation in oxygen availability, and Gilman et al. (1987) note, “…woody plants respond to compacted soils by producing a shallow root system. … Roots which develop in well-aerated soil are long and light in color with many root hairs. Those with insufficient O2 are thicker, twisted, shorter, darker in color and have few root hairs. Soil aeration affects root distribution by restricting deeper penetration at low O2 concentrations. Frequently in low O2 environments, a dense mat of roots develop within the top several cm of soil” (Gilman, Leone and Flower, Effect of soil compaction and oxygen content on vertical and horizontal root distribution 1987).
The general size and shape of the soil particles affects the distance between particles. The general amount of the soil particle mass (total weight of soil particles) per unit volume is referred to as the “bulk density” of a soil. As the soil becomes more compacted, the bulk density goes up. To help visualize how particle size and shape affect bulk density and resultant soil void space, imagine filling a bathtub with open umbrellas. The irregularly-shaped umbrella (e.g. particles) would not fit together very well and the large size would provide a plenty of mechanically-separated space in between them. If this bathtub was then filled with air or water, there would be a large pockets of space or capacity for oxygen or moisture. The “uncompacted” spaces between the particles would allow for the adventitious growth of roots. Now envision that same bathtub filled with marbles. The smaller, regularly-shaped particles fit together more tightly and would contain less space between them. They also would have less volume for root-available moisture and oxygen due to the smaller voids between particles, and growth of roots in this material would require more displacement of densely-packed particles with less nutrient availability.
Soil compaction, or relative bulk density, can be used as a proxy for chemical/nutrient availability, and mechanical separation of pores for adventitious rooting is one of the most important things to consider in examining the role of the structural soils or rock-based soil. As compared to a standard loam planting soil, structural soils and clear stone both have a relatively-larger particle size that mechanically maintains the larger void space between adjacent particles, even under compaction to structural loading conditions. The only difference between structural soils and clear stone is the addition of smaller particles and organic material in the structural soils as a component of the larger particles that maintains the overall soil structure. This additional material in the structural soil somewhat reduces the overall volume of the pore space, but adds some additional plant-available water and nutrient content to the soil that the clear stone would not offer.
Designers should always consider the natural history and the preferred conditions in which the tree developed before specifying its planting condition. Large volumes of uncompacted native soil are usually best. Under the right conditions, adventitious rooting can occur in structural soils and ballast. A material that may limit the growth of the chosen species (for example, specifying an acidic species in a basic/alkali soil media) will not produce a healthy tree, no matter the soil oxygen content.
Andrea Wedul is a landscape architect with The Kestrel Design Group.
Literature Cited
Bartens, J., P. E. Wiseman, and E. T. Smiley. “Stability of landscape trees in engineered and conventional urban soil mixes.” Urban Forestry & Urban Greening 9 (2010): 333-338.
Coder, Kim D. December 2007. Soil Compaction, Stress and Trees: Symptoms, Measures,Treatments. University Of Georgia Warnell School Of Forestry & Natural Resources. Warnell School Outreach Monograph WSFNR07-9. Available October 2013 from University of Georgia.
Day, S. D., and N. L. Bassuk. “A review of the effects of soil compaction and amelioration treatments on landscape trees.” Journal of Arboriculture 20 (1994): 9-17.
Di Iorio, A., B. Lasserre, L. Petrozzi, G. S. Scippa, and D. Chiatante. “Adaptive longitudinal growth of first-order lateral roots of a woody species (Spartium junceum) to slope and different soil conditions – upward growth of surface roots.” Environmental and Experimental Botany 63 (2008): 207-215.
Gilman, E. F. “Deflecting roots near sidewalks.” Arboriculture & Urban Forestry (ISA), 2006: 18-22.
Andreal Wedul is a landscape architect with The Kestrel Design Group.
Gilman, E. F., I. A. Leone, and F. B. Flower. “Effect of soil compaction and oxygen content on vertical and horizontal root distribution.” Journal of Environmental Horticulture 5, no. 1 (1987): 33-36.
Lindsey, P., and N. Bassuk. “Redesigning the urban forest from the ground below: a new approach to specifying adequate soil volumes for street trees.” Arboricultural Journal 16 (1992): 25-39.
Lynch, J., P. Marschner, and Z. Rengel. “Effect of Internal and External Factors on Root Growth and Development.” In Marschner’s Mineral Nutrition of Higher Plants (3rd Edition), by P. (Ed.) Marschner, edited by P. Marschner, 331-346. Waltham, MA: Academic Press – Elsevier Ltd., 2012.
McMichael, B. L., and J. E. Quisenberry. “The impact of the soil environment on the growth of root systems.” Environmental and Experimental Botany 33 (1993): 53-61.
Postma, J. A., U. Schurr, and F. Fiorani. “Dynamic root growth and architecture responses to limiting nutrient availability: linking physiological models and experimentation.” Biotechnology Advances xxx (In Press) (2013): xxx-xxx (12).
Yau, Peter. “Tree root damages to infrastructure study commissioned by Westpool Group NSW*.” 2012. (accessed November 14, 2013).
Top image Flickr credit: born1945





It is important to understand what constitutes a “positive” response by the tree, as seen by the growth of roots. Growth response triggers may be chemical, physical, or biological in nature. A tree’s growth and development is entirely directed towards positive responses to environmental stimuli. If there is soil-available moisture, stomata open and photosynthesis initiates.